Intrauterine Surgery

Oxiplex/IU Adhesion Barrier Gel is a unique formulation of the company’s proprietary biomaterial. It is a flowable gel that is intended to serve as a temporary, absorbable mechanical barrier separating surgically traumatized opposing tissue surfaces in the uterine cavity where adhesions could potentially form. Oxiplex/IU is supplied sterile and ready to use in one 10 mL syringe with a sterile applicator.
Intrauterine Adhesions
Intrauterine (IU) adhesions are bands of scar tissue that form between the inner walls of the uterus, resulting from trauma to the uterus. Any surgical intervention of the uterine cavity can potentially create adhesions. Adhesions can occur even when surgeons use the best techniques, but using mechanical barriers to separate traumatized tissues during the healing period can reduce postoperative adhesions.
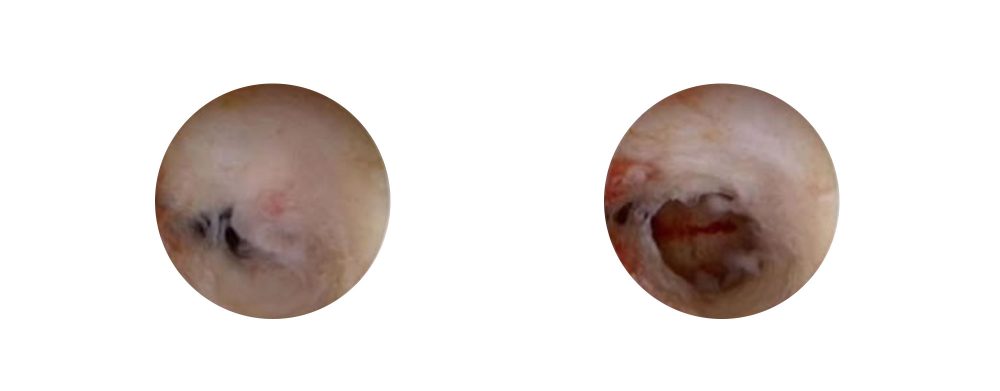
Clinical Consequences of Intrauterine Adhesions
Many women develop IU adhesions following intrauterine surgery. Although some intrauterine adhesions can be asymptomatic, they can also cause problems such as:
- Infertility
- Menstrual cycle abnormalities
- Pain and discomfort
- Dysmenorrhea
- Recurrent pregnancy loss
Advantages
The advantages of using Oxiplex/IU Adhesion Barrier Gel in intrauterine surgery include:
Benefits
- Exceptional safety
- Designed and indicated for use in intrauterine surgery
- Ready to use
- Fast application, thorough coverage
- Colorless
Protects the Procedure
- Separates and coats tissues
- Reduces adhesions
Optimizes Healing
- Re-operations can be complicated by adhesions at original operative site
Improves Outcomes
- Less de novo (i.e., new) adhesions
How it Works
We designed Oxiplex/IU gel to coat surfaces that are damaged in the uterine cavity during intrauterine surgery. Application of the gel forms a temporary barrier as tissue heals. This protective physical barrier keeps opposing tissues separated, allowing them to heal optimally. Clinical studies have shown this protective barrier reduces the number of de novo adhesions following hysteroscopic surgery.1,2,3
Adhesion barriers are a proven method of enhancing good surgical technique for procedures such as:
- Adhesiolysis
- Treatment for Asherman’s syndrome
- Dilation and Curettage
- Removal of retained products of conception
- Myomectomy
- Polypectomy
- Uterine septum surgery
Application of Oxiplex/IU gel during surgery is simple and takes only seconds. After surgery, the gel is absorbed by the body over a short period of time.
Indication
Oxiplex/IU is intended to be used as an adjunct to intrauterine procedures for reducing the incidence, extent, and severity of adhesions.
Clinical Experience
Exceptional Clinical Results
DiSpiezio Sardo et al used Oxiplex/AP following hysteroscopic metroplasty to expand dysmorphic uteri in a prospective, observational study in 30 infertile women.
- Oxiplex/AP gel was applied into the uterine cavity at the completion of the procedure through the inflow channel of the hysteroscope.
- At the mean follow-up of 15 months, clinical pregnancy rate was 57% and term delivery rate was 65%.
- Low incidence of post surgical synechiae attributed to the use of Oxiplex/AP gel.
Di Spiezio Sardo et al used Oxiplex/AP following hysteroscopic surgery in a randomized study of 110 patients.
- Follow-up at one month showed that Oxiplex/AP treated patients had fewer ‘de novo’ adhesions (6% vs 22%) compared to control.
- Patients treated with Oxiplex/AP also demonstrated improvement in the degree of patency of the internal uterine osteum.
Fuchs et al. used Oxiplex/AP following hysteroscopic surgery in a randomized study of 52 women.
- Follow up at 20 months showed that Oxiplex/AP treated patients had fewer moderate to severe intra-uterine adhesions (4% vs 14%) compared to control.
- Patients treated with Oxiplex/AP had an improved fertility rate (27% vs 14%) compared to controls.
Case Studies
Instructions For Use
Click here to request the most recent revision of this product’s instructions for use. Please state the product name for the IFU requested in the comments section.
Availability
FzioMed offers innovative adhesion barrier products such as Oxiplex®, Oxiplex/SP® Gel, Interpose®, MediShield™, Oxiplex/AP®, Oxiplex/IU®, and DYNAVISC®. These products are designed for use by medical professionals. Click here to be contacted by a distributor in your country.
Last Updated: March 5, 2024
