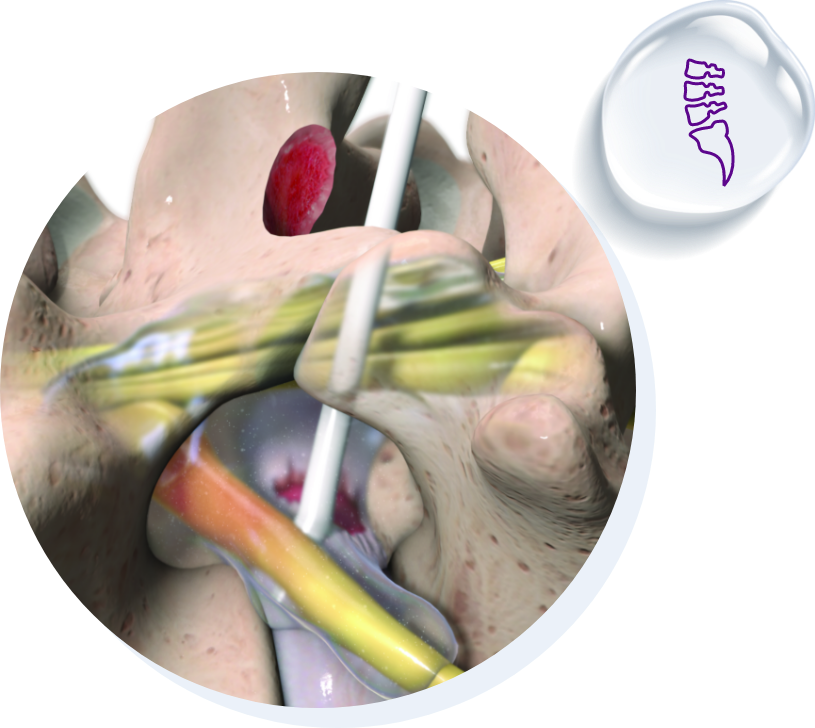
Oxiplex is an FDA De Novo device. It is a synthetic, absorbable gel intended as an adjunct to lumbar spinal surgery in adult patients with leg pain, back pain, and neurological symptoms undergoing discectomy to reduce leg pain and neurological symptoms. Oxiplex should only be used after hemostasis during wound closure in adult patients. It is supplied sterile, for single use only, should not be re-used or sterilized, and should not be used in the presence of infection. Healthcare professionals should refer to the Instructions for Use for a complete list of Contraindications, Warnings and Precautions.
Caution: Federal law restricts this device to sale by or on the order of a physician.
References
-
Fischgrund J, et al. Dual-Polymer Carboxymethyl Cellulose and Poly(Ethylene Oxide) Gel Reduces Leg and Back Pain in Patients With Severe Leg and Back Pain Following Single-Level Partial Discectomy. Spine 2026;51:384-392.
- Rhyne A, et al. Oxiplex reduces leg pain, back pain and associated symptoms following lumbar discectomy. Spine 2012;37:631-641.
- Assietti R, et al. Use of carboxymethylcellulose/polyethylene oxide gel in microdiscectomy with interlaminectomy, a case series comparison with long-term follow-up. Spine 2008;33:1762-1765.
*Compared to surgery alone at 6 months in patients with severe leg and back pain prior to surgery.